What is Lean?
Are you an experienced chemist frustrated by how chaotic your day-to-day work in the laboratory is? Or perhaps you are a recent graduate who has just joined your first laboratory job in the industry setting and heard the terms Lean, 5S, or Kaizen but don't fully understand what they mean? You have come to the right place.
Lean is a philosophy and a set of practices focused on maximizing value while minimizing waste. It is about doing the minimum necessary to achieve the optimal result and maximizing the flow of your process. It has many definitions and is often branded as something mysterious or hard to grasp- However, in reality, it is anything but that.
The concept of Lean originated in Japan after World War II. It grew out of the Toyota Production System (TPS), developed by Taiichi Ohno and Shigeo Shingo at Toyota in response to the scarcity of resources and capital. This necessity forced Toyota to develop a system in which mass production shifted toward the total elimination of waste and the creation of a pull system, where parts are replenished only as they are consumed.
Let’s dig deeper.
“Lean is about doing the absolute minimum necessary to get the desired result.”
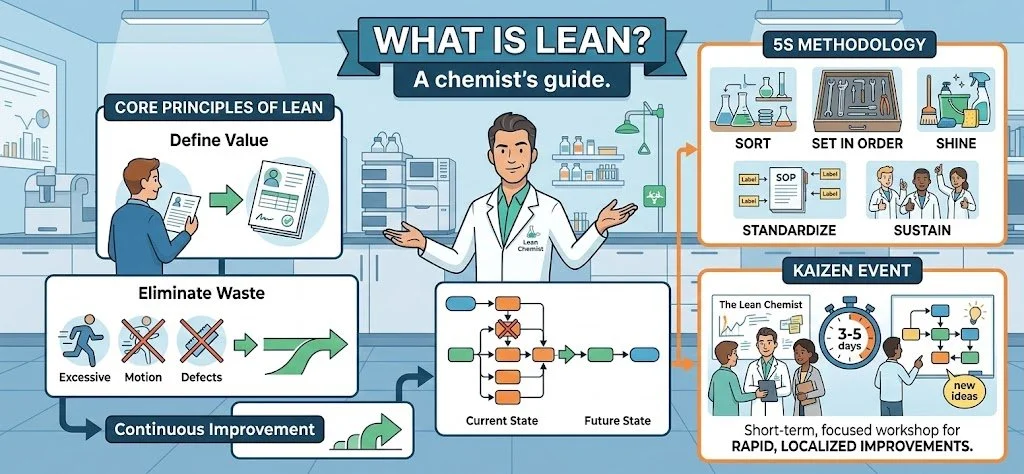
Chapter 1. Core Principles of Lean
To make it simple, there are five core principles of Lean you should keep in mind.
Define what value is. In a laboratory context, value is defined by what your customer needs from you. Most likely, they provide a sample and want specific analysis results (purity, concentration, or composition). Beyond the data, they want it fast, in an easily digestible format, and with 100% accuracy. Anything that doesn't contribute to these three goals is technically waste.
Map the value stream. What is a value stream in a lab? It is the entire path a sample takes from arrival to the delivery of the final report. This path can be broken down into dozens of steps. Some add value; many do not. Identifying these non-value-adding steps is the first step toward eliminating waste.
Create Flow. Ideally, a sample should move through your process without stopping. In a perfect world, an analyst immediately begins sample prep, moves to the equipment (e.g. HPLC, GC, AAS), and runs the analysis. The equipment prints the report, the analyst reviews it, and you send it to the client. Voila! In reality, obstacles are everywhere: a backlog of twenty other samples, a 12-hour dissolution time, or standards that fall out of specification. When the sample stops moving, flow is broken.
Establish pull. Imagine the week after Christmas. Your sample cabinet is empty. A single sample arrives for HPLC analysis. Your analyst is ready, but you tell them to wait until the autosampler is full to be efficient. Wrong. According to Lean, waiting for a full batch creates a bottleneck. You leave the chemist idle, only to later overwhelm them with twenty results to review at once. If there’s an error in the reagent prep, you’ve now ruined thirty samples instead of catching the mistake after one. This kind of batching kills your Turnaround Time (TAT) and provides zero value to the customer waiting for that first sample.
Seek perfection. Have you ever heard that a process is good enough or this is how we’ve always done it? That is a disaster waiting to happen. In a competitive industry, you must constantly evolve. Seeking perfection doesn't mean expecting your team to never make a mistake. It means scrutinizing the process. If a sample prep procedure has thirty steps or an SOP is forty pages long, an error isn't the fault of the analyst. It’s the fault of an inefficient process.
Alright, you probably get the gist of it. Now, let’s move into waste.
Chapter 2. What is Waste?
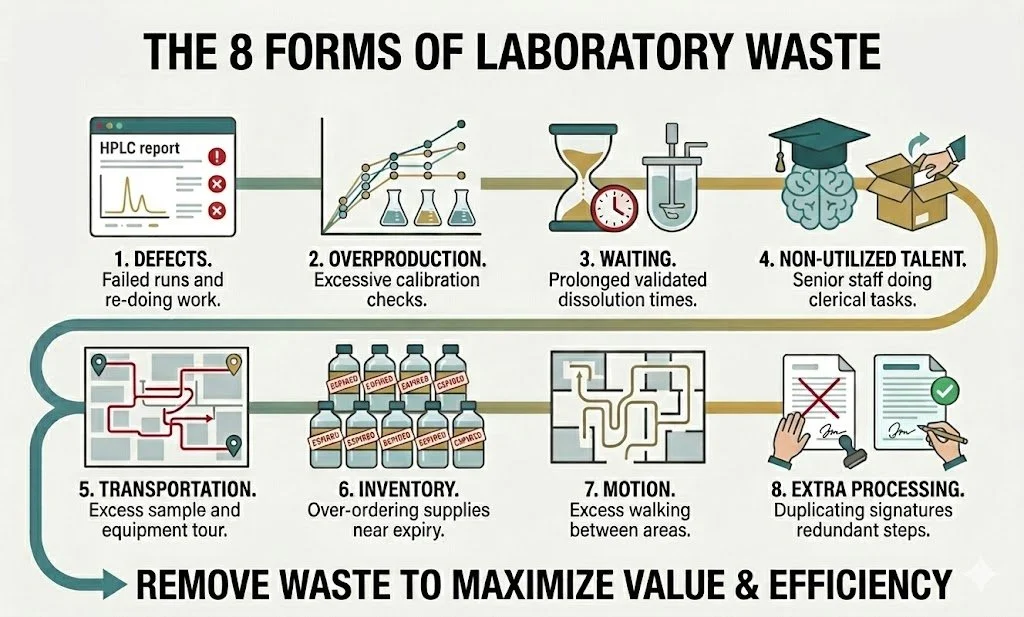
The elimination of waste is a core tenet of Lean. In a laboratory, waste typically falls into these eight categories:
Defects: Defects are anything not done right the first time. In a lab, this could be a failed HPLC run due to an expired mobile phase, a wrongly diluted sample, or a transcription error on a report. Every defect requires rework, which consumes time and reagents without adding value.
Overproduction: This occurs when you do more work than is actually required. For example, your procedure might require a full 5-point calibration curve and injections in triplicate, even though your quality system or regulatory requirements only necessitate a single-point calibration check to remain in compliance.
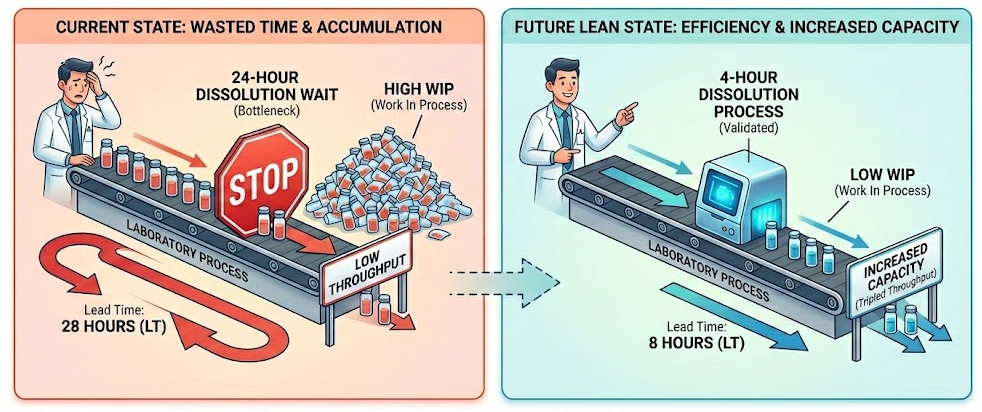
Waiting: Waiting is often the most visible waste. Imagine a validated procedure that requires 24 hours for sample dissolution, even though the sample is perfectly dissolved in five minutes. This happened because the dissolution time wasn't properly optimized during method development to "save time." Now, that wasted 19 hours repeats every single day for the next ten years.
Non-utilized talent: This is the waste of human potential. If your senior chemist with a PhD is spending their morning opening shipping packages instead of focusing on the method development and validation you hired them for, you are wasting the very expertise the company pays for.
Transportation: Transportation adds zero value; a sample doesn’t get "better" because it took a tour of the building. If you have to collect specialized glassware from central storage on one side of the facility, move samples to a weighing room in the basement, and then transport them back to the main lab for digestion, you are observing pure transportation waste.
Inventory: Inventory waste is often disguised as being prepared. A technician warns that solvent is low, so you order forty bottles instead of twenty just in case. When they arrive, they fill the cabinet to the brim, only for you to realize the batch expires in two months. You’ve now wasted money and storage space on chemicals you can't even use.
Motion: While transportation refers to the movement of samples or tools, motion refers to the movement of people. If your sample cabinet is on the first floor, your balance is on the second, and your HPLC is back on the first, your analysts are spending more time walking than they are doing science. It might be good for their daily step count, but it creates zero value for the customer.
Extra processing: Extra processing is doing more work than the quality standard requires. Consider a GMP laboratory where you print an FTIR report, sign it manually, scan it into a system, and then sign it again electronically because two different SOPs overlap. If you have a 21 CFR Part 11 compliant system with an audit trail, these manual steps are redundant. You should be building quality into the process by connecting the system directly to your electronic records.
By the way, this spells out as DOWNTIME. Easy to remember, right?
As you can see, there are various forms of waste. Before we get into it deeper, lets looks at actually why removing waste is essential.
Chapter 3. Benefits of Lean
By now, you understand the core principles of Lean and what waste actually looks like. Hopefully, these examples have shown you how logical Lean can be: in short, don't do useless stuff. However, that is often easier said than done. Laboratories are complex beasts with hundreds of analytical methods and workloads that never stop piling up. It feels like nobody has time to do anything "extra". but that is the ultimate Lean fallacy.
Let’s look deeper at the 24-hour dissolution example from earlier. Your analysts, who have years of experience, are convinced the samples dissolve in just a four hours. They share this insight with you. You recognize that waiting is a form of waste, but you frown; changing a validated analytical method under strict regulatory requirements is notoriously difficult. You thank them for their observation and promise to look into it.
Then, the day vanishes in a blur of back-to-back meetings. Two weeks pass. You hear your analysts sighing as you walk by; the workload is piling up, and your Turnaround Time (TAT) is at an all-time high. Frustrated, you meet with management to ask for more headcount. Your manager counters: “Hiring is on hold. You need to improve your team’s performance with the resources you have.”
You walk back to your desk with that challenge ringing in your ears. It sounds like a corporate cliché until you remember that 24-hour dissolution step. If your team is right, you aren’t just waiting on chemistry: you’re wasting time just because the SOP says so.
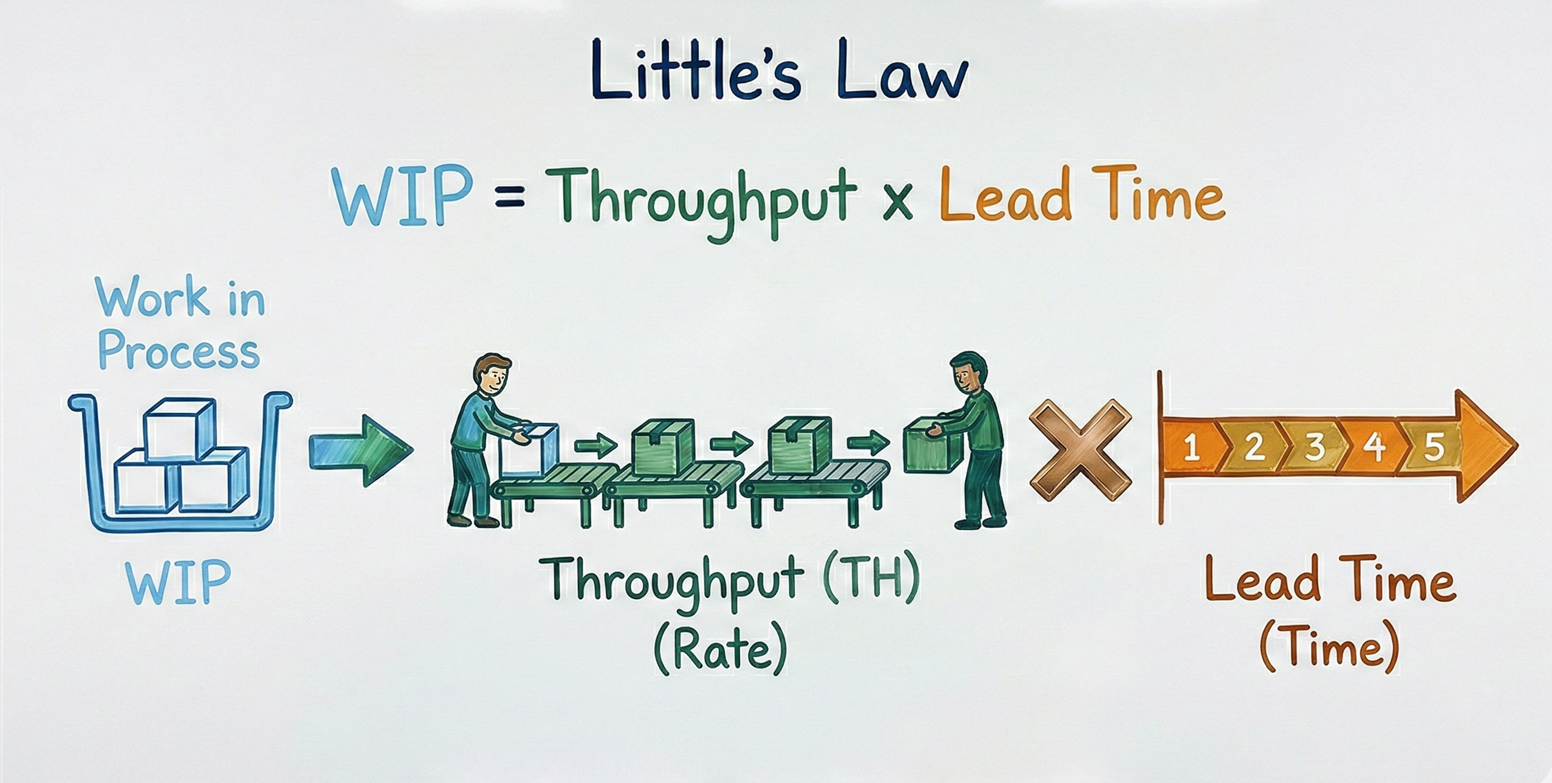
To convince the regulatory team to re-validate the method, you need more than a hunch; you need a business case. You sit down with Excel to calculate your Lead Time (LT), Throughput (Th), and Work in Process (WIP) using a principle called Little’s Law.
In this context,
WIP = The number of samples currently stuck in the system.
Throughput = The rate at which samples are completed (e.g., samples per hour, day or week).
Lead Time = The total time a sample spends in the lab, from login to final report.
The point of Little’s Law is that you can’t change one of these categories without affecting the others. For your laboratory, this means:
If you want to reduce WIP, you must work faster (e.g. reduce Lead Time).
If the Lead Time of your laboratory increases, but samples keep arriving at the same rate (Throughput), your WIP will increase.
Get it? Alright, let’s move back to our earlier example.
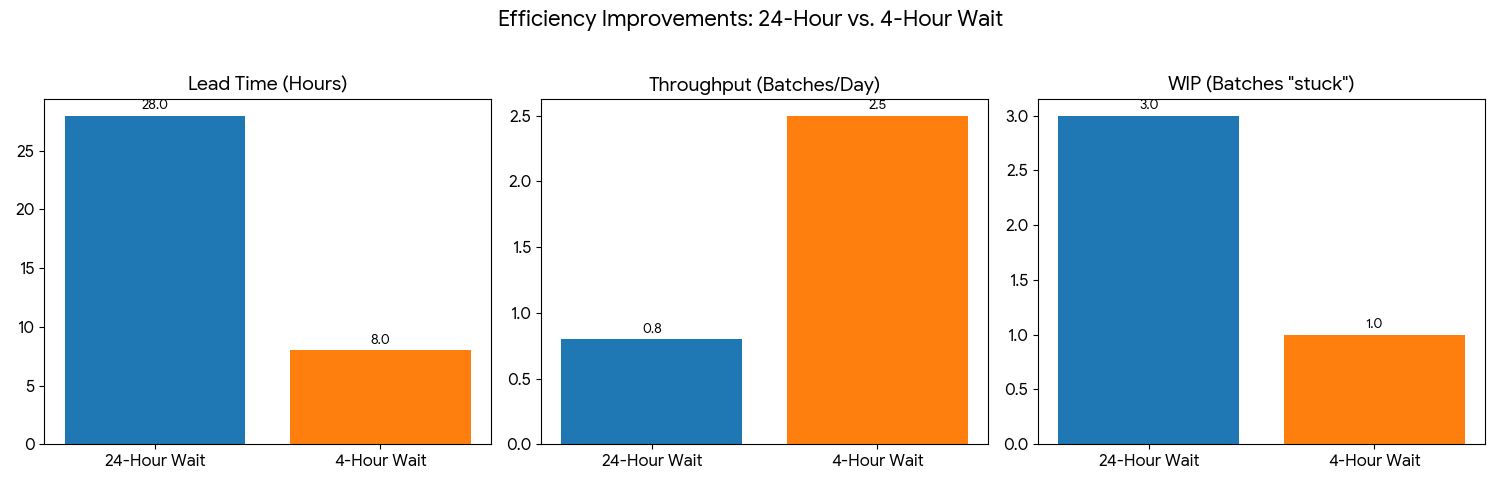
Assuming a standard batch of 20 samples:
Current State: 2 hours of prep + 24 hours waiting + 2 hours analysis = 28 hours total.
Future State: 2 hours of prep + 4 hours waiting + 2 hours analysis = 8 hours total.
If you run these calculations based on a standard work week, the shift is staggering:
“Look at this,” you point to the 71 % reduction in Lead Time. “By cutting the waste in waiting for the sample to dissolve, we triple our theoretical capacity. Right now, we are paying for 20 hours of samples just sitting on a shelf doing nothing.”
One chemist speaks up: “But the validation work will take forty man-hours to prove the 4-hour mark is equivalent.”
“True,” you respond, “but if we spend 40 hours now to save 20 hours per batch, we break even after just two batches. After that, every single day is pure profit in time.”
The frustration in the room shifts toward focus. The team realizes they aren't too busy to improve; they are too busy because they haven't improved. You decide to run a small pilot study to gather the data needed to trigger a formal change control.
Alright, so now you understand the core principles of Lean and what waste actually is. You have seen an example of what the benefits can actually mean for your laboratory. Now, let’s look at what the literature says and some actual data that’s available.
Chapter 4. Literature review
Example 1:
In a study by Inalal. (2017), researchers applied Lean Six Sigma (DMAIC) to a clinical laboratory and achieved a dramatic reduction in Turnaround Time (TAT) by targeting the hidden waste in the pre-analytical phase. By streamlining the sample reception area and eliminating 3 hours and 22.5 minutes of non-value-adding work, they saw STAT sample TAT drop from 68 to 59 minutes. Beyond speed, the Lean intervention successfully reduced steps prone to medical errors and biological hazards from 30% down to 3%, proving that removing waste improves both safety and efficiency.
Example 2:
A systematic review published in PLOS One by Cherie et al. (2024), several benefits of Lean implementations were found in the explored studies:
Implementing Lean principles in routine laboratory testing resulted in an overall 76.1% reduction in laboratory TAT
In one laboratory, the mean TAT for creatinine tests dropped from 54 minutes to 23 minutes (a 57.4% reduction) after eliminating wasteful walking patterns and instrument downtime.
Lean interventions led to a 47.2% reduction in hematology TAT (from 179.4 to 94.7 minutes) and a 22.3% reduction in biochemistry TAT for a pathology department
In emergency settings, Lean methodology reduced TAT for chemistry by 35.4% and urinalysis by 61.4% by removing technician distractions and optimizing sample collection.
Beyond speed, Lean increased staff productivity by up to 20% and improved the overall quality of care and patient satisfaction by providing faster diagnostic results.
Example 3:
In a study by Gupta et al. (2018) Lean methodology was utilized to reduce turnaround time (TAT) in a clinical laboratory at a super-specialty hospital in India. Researchers utilized specialized Lean tools, including Value Stream Mapping (VSM), Gemba walks, and Root Cause Analysis, to identify and eliminate non-value-added steps in the testing process. A key finding was the identification of 17 major bottlenecks across the hematology and biochemistry departments that were significantly limiting laboratory throughput. As a concrete result, the average TAT in the hematology laboratory was reduced from 180 minutes to 95 minutes after a four-month research intervention. The biochemistry laboratory also saw success, with its TAT decreasing from 268 minutes to 208 minutes, ultimately leading to improved patient satisfaction and increased hospital revenue.
Example 3:
In pharmaceutical industry, Byrne et al. (2021) implemented a customized 7-step Lean Six Sigma (LSS) framework to address production backlogs at a paracetamol manufacturing site during the COVID-19 pandemic. The study focused on reducing packaging line downtime, which had become a critical bottleneck due to an unprecedented surge in demand for pain relief medication. By utilizing tools like Value Stream Mapping (VSM) and DMAIC, the team successfully identified and eliminated the root causes of "short stops" and equipment breakdowns on the production lines.
The LSS project successfully eliminated the specific packaging line "short stops" and downtime issues that were causing production delays. The intervention resulted in documented savings of just under $500,000 for the manufacturing facility.
“The intervention resulted in documented savings of just under $500,000 for the manufacturing facility.”
Chapter 5. The Toolkit for Process Improvement
In the literature review earlier, I showed you some benefits of Lean for turnaround time, reduction in workload or actual monetary savings. I mentioned plenty of methods the articles utilized. Most of which, we have not explored. Here are some of these tools you can use to actually reduce waste and improve your processes:
Value Stream Mapping (VSM): This visual tool maps out every process step to distinguish between value-adding and non-value-adding activities from the customer's perspective. It allows laboratories to pinpoint exactly where bottlenecks occur, serving as a blueprint for a more efficient future state.
Lean Six Sigma (LSS): This methodology combines waste elimination with a data-driven approach to reduce process variation across five structured phases: Define, Measure, Analyze, Improve and Control (DMAIC). It is designed to solve complex problems by identifying and controlling unknown root causes.
Plan-Do-Check-Act (PDCA): This four-step iterative cycle involves planning a change, executing a pilot, checking the data for equivalence, and acting to standardize the workflow. It ensures that laboratory improvements are sustained through a continuous cycle of testing and validation.
Gemba Walks: his technique involves leaders physically visiting the "actual place" of work to observe real-time operations. By observing the actual workflow, teams can identify hidden inefficiencies like excessive technician movement or disorganized workspaces.
Root Cause Analysis (RCA): This systematic process identifies the underlying factors of a problem rather than just treating its superficial symptoms. Tools like the "5 Whys" or Fishbone diagrams are utilized to ensure permanent solutions are implemented to prevent reoccurring delays.
Kaizen event: This short-term, intensive workshop brings cross-functional teams together to improve a specific process over three to five days. It aims to implement rapid, localized solutions that achieve measurable improvements in laboratory productivity or quality.
5S Methodology: This workplace organization tool stands for Sort, Set in order, Shine, Standardize, and Sustain. By creating a highly organized and standardized environment, laboratories can reduce the "motion waste" of technicians searching for reagents or tools, directly impacting speed and safety.'
Kanban: This visual management system uses signals to control the flow of samples and limit work in progress. It prevents batches from getting "stuck" by ensuring the lab only starts new work when there is capacity to finish it.
Poka-Yoke: This error-proofing method implements fail-safes, such as color-coding or software blocks, to prevent human mistakes during prep. It protects throughput by ensuring the 8-hour lead time isn't derailed by the need for rework.
Heijunka: This tool levels out the production schedule to eliminate the "peaks and valleys" of daily laboratory sample arrival. By creating a steady workload, it prevents the team from feeling overwhelmed and helps maintain a consistent pace.
I know, that’s a lot to pack! We won’t have time to dig deeper into these tools in this article, as each of these tools would take an entire article to fully explain. In fact, that is exactly what I am planning on doing in my next articles: digging deeper on how to actually improve performance in your laboratory.
Stay tuned for more!
Sources:
Earley, J.A.A. (2016) The lean book of lean: a concise guide to lean management for life and business. Chichester: John Wiley & Sons.
Inal, T. C., Goruroglu Ozturk, O., Kibar, F., Cetiner, S., Matyar, S., Daglioglu, G. and Yaman, A. (2018) ‘Lean six sigma methodologies improve clinical laboratory efficiency and reduce turnaround times’, Journal of Clinical Laboratory Analysis, 32(1), p. e22180. doi: 10.1002/jcla.22180.
Cherie, N., Berta, D.M., Tamir, M., Yiheyis, Z., Angelo, A.A., Tarekegn, A.M., Chane, E., Nigus, M. and Teketelew, B.B. (2024) 'Improving laboratory turnaround times in clinical settings: A systematic review of the impact of lean methodology application', PLOS ONE, 19(10), p. e0312033. doi: 10.1371/journal.pone.0312033.
Gupta, S., Kapil, S. and Sharma, M. (2018) ‘Improvement of laboratory turnaround time using lean methodology’, International Journal of Health Care Quality Assurance, 31(4), pp. 295–308. doi: 10.1108/IJHCQA-08-2016-0116.
Byrne, B., McDermott, O. and Noonan, J. (2021) ‘Applying Lean Six Sigma Methodology to a Pharmaceutical Manufacturing Facility: A Case Study’, Processes, 9(3), p. 550. doi: 10.3390/pr9030550.
The visualizations in this article were generated using user prompts by Google Gemini (2026) based on the factual content in the article.